G-Protein Pathway-
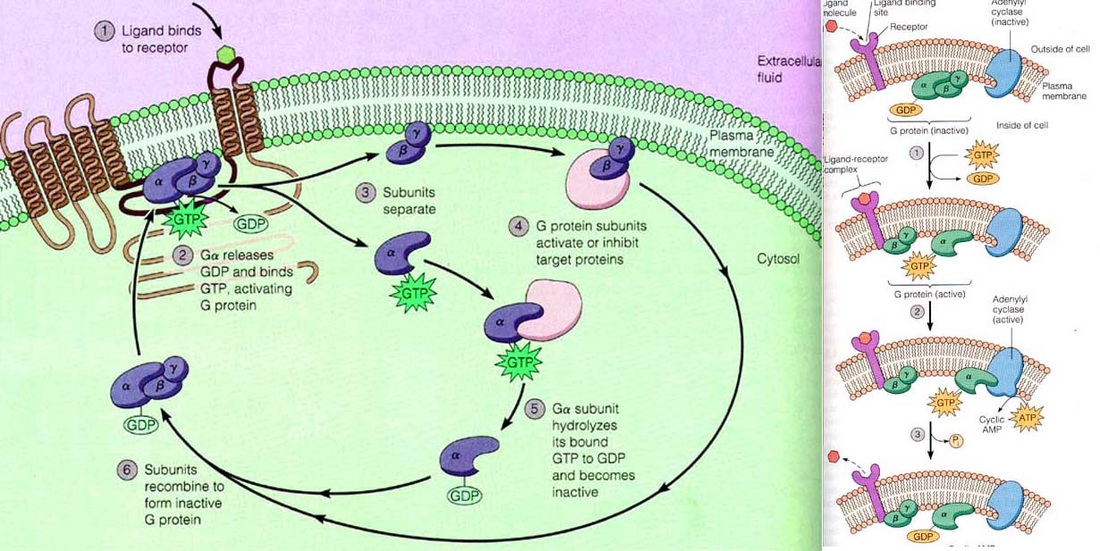
The heterotrimeric guanosine, triphosphate, nucleotide–binding proteins (G proteins) are signal
transducers that communicate signals from many hormones, neurotransmitters, chemokines, and autocrine and paracrine factors. The extracellular signals are received by members of a large superfamily of receptors with seven membrane-spanning regions that activate the
G proteins, which route the signals to several distinct intracellular signaling pathways. These pathways interact with one another to form a network that regulates metabolic enzymes, ion
channels, transporters, and other components of the cellular machinery
controlling a broad range of cellular processes, including transcription, and secretion. These cellular
processes in turn regulate systemic functions such as embryonic development, learning and memory, and organismal homeostasis.
Tyrosine Kinase Pathway-
The receptor tyronsine kinase (RTK) influence the cell cycle, cell migration, cell metabolism, and cell proliferation--virtually all aspects of the cell are affected by signaling through these receptors. Alterations to the function of these receptorsand their signaling pathways can lead to cancer in humans and other animals.
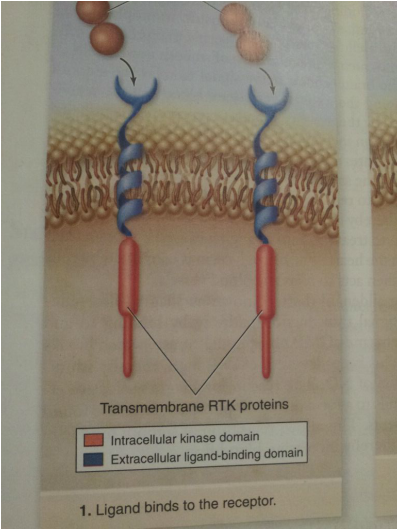
Activation of a Receptor Tyrosine Kinase (RTK):
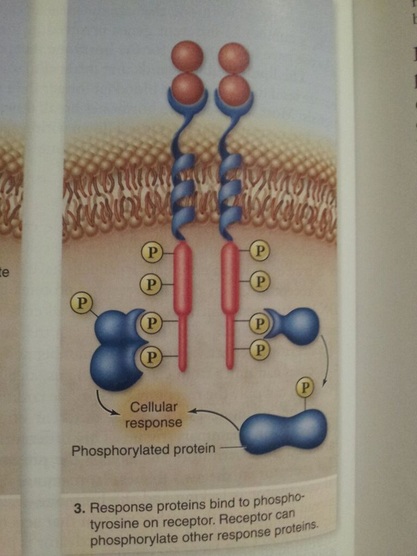
These membrane receptors bind hormones or growth factors that are hydrophilic and cannot cross the membrane. The receptor is a transmembrane protein with an extracellular ligand binding domain and an intracellular kinase domain. Signal transductionpathways begin with response proteins binding to phosphotyrosine on receptor, and by receptor phosphorylation of response proteins.
Structure:
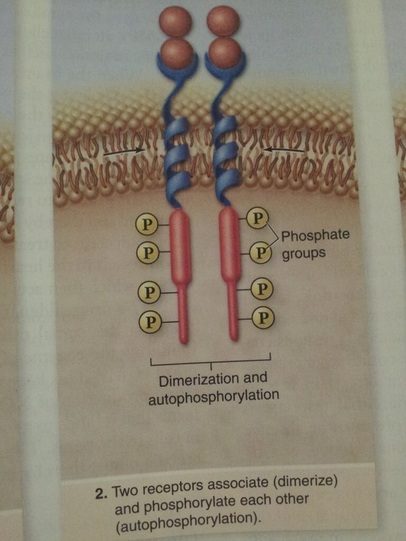
Receptor tyronsine kinases have a relatively simple structure consisting of a single transmembrane domainthat anchors them in the membrane, an extracellular ligand-binding domain, and an intracellular kinase domain. The kinase domain contains the catalytic site of the receptor, which acts as a protein kinase that adds phosphate groups to tyrosines. On ligand binding to a specific receptor, two of these receptor-ligand complexes associate together and phosphorylate each other, a process called autophosphorylation.
Knowledge Extension:
Proteins called scaffold proteins are thought to organize the components of a
kinase cascade into a single protein complex, the ultimate in a signaling
module. The scaffold protein binds to each kinase in the cascade, organizing them so each substrate is next to its enzyme.This organization also sequesters the kinases from other signaling pathways in the cytoplasm.
The link between RTK and the MAP kinase cascade is a small GTP- binding protein (G-protein) called Ras.
Activation of a Receptor Tyrosine Kinase (RTK):
These membrane receptors bind hormones or growth factors that are hydrophilic and cannot cross the membrane. The receptor is a transmembrane protein with an extracellular ligand binding domain and an intracellular kinase domain. Signal transductionpathways begin with response proteins binding to phosphotyrosine on receptor, and by receptor phosphorylation of response proteins.
Structure:
Receptor tyronsine kinases have a relatively simple structure consisting of a single transmembrane domainthat anchors them in the membrane, an extracellular ligand-binding domain, and an intracellular kinase domain. The kinase domain contains the catalytic site of the receptor, which acts as a protein kinase that adds phosphate groups to tyrosines. On ligand binding to a specific receptor, two of these receptor-ligand complexes associate together and phosphorylate each other, a process called autophosphorylation.
Knowledge Extension:
Proteins called scaffold proteins are thought to organize the components of a
kinase cascade into a single protein complex, the ultimate in a signaling
module. The scaffold protein binds to each kinase in the cascade, organizing them so each substrate is next to its enzyme.This organization also sequesters the kinases from other signaling pathways in the cytoplasm.
The link between RTK and the MAP kinase cascade is a small GTP- binding protein (G-protein) called Ras.
The Insulin Receptor-
The insulin receptor is a receptor tyrosine kinase that initiates a variety of of cellular responses related to glucose mataboism. One signal transduction pathway that this receptor mediates leads to the activation
of the enzyme glycogen synthase. This enzyme converts glucose to oxygen.
Maintaining Homeostasis-
Insulin is essential for maintaining glucose homeostasis and regulating carbohydrate, lipid, and protein metabolism. Insulin is a reeptor tyrosine kinase, this shows how important this pathway is to homeostasis
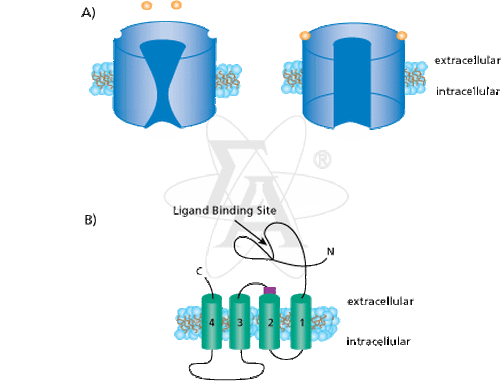
Ligand-gated Ion Channel-
Chemically gated ion channels are receptor proteins that allow the passage of ions. The receptor proteins that bind many nerotransmitters have the same basic structure. Each is a membrane protein with multiple transmembrane domains, meaning that the chain of amino acids threads back and forth across the plama membrane several times. In the center of the protein is a pore that connects the extracellular fluid with the cytoplasm. The pore is big enough for ions to pass through, so the protein functions as an ions channel. An example of a ligand-gated ion channel would be neurons. Neurons are molecular gates triggere chemically to open or close.
Structure- The structure can be defined as a multipass transmembrane protein forminga central pore.
Structure- The structure can be defined as a multipass transmembrane protein forminga central pore.
Maintaining Homeostasis-
Ligand- gated ion channels help maintain homeostasis by regulation the number of ions in the cell. Mutations in ion channels can lead to protein misfolding and extensive ER-associated degradation (ERAD), causing ion channel diseases, such as epilepsy.